The Relationship Between Cortisol and Estrogen in Perimenopause and PCOS
By Dr. Fiona McCulloch, ND, Board Certified in Functional and Naturopathic Endocrinology, with 25+ years in clinical practice
Cortisol and estrogen affect each other bidirectionally, in ways that are more specific than most online resources describe. Chronic stress can suppress estrogen production through a defined brain pathway (GnRH). Estrogen, in turn, changes how cortisol appears on blood tests: oral estrogen raises total cortisol readings by approximately 67%, even when active cortisol is unchanged (1). In perimenopause and PCOS, these interactions produce distinct, identifiable symptom patterns that are often misattributed to stress or mood disorders.
When Stress Symptoms Feel Physical, Not Psychological
If your stress tolerance has changed, if you feel more reactive, more fatigued in the mornings, or more wired at night than you used to, there is a physiological explanation worth understanding. In perimenopause especially, many patients describe a specific kind of anxiety that feels very physical: heart racing, fearfulness, difficulty breathing, feeling hot and itchy and anxious. This pattern is distinct from psychological overwhelm. It feels different because it is different: the cortisol and estrogen systems are driving it through mechanisms that have nothing to do with whether you are “too stressed.”
These symptoms are often normalized, attributed to general stress or classified as anxiety or depression, particularly when they appear without an obvious external cause. Understanding the hormonal mechanism behind them can help clarify whether a clinical assessment makes sense for your situation.
How Chronic Stress Lowers Estrogen Production
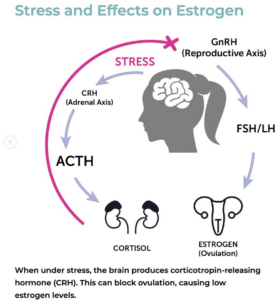
Severe chronic stress can block ovulation by affecting a brain hormone called GnRH (gonadotropin-releasing hormone) (2). Here is the mechanism: when stress is sustained, cortisol production remains elevated. Elevated cortisol signals the hypothalamus to reduce GnRH release. Because GnRH controls the release of LH and FSH, the hormones that direct ovarian function, and reduced GnRH means reduced estrogen production. Since estrogen is made as follicles prepare for ovulation, this suppression can disrupt the entire ovulatory cycle.
This is not a malfunction. It is the body’s way of deprioritizing reproduction under sustained physiological threat, an energy conservation response. But the clinical result can be confusing: irregular cycles, fatigue, low mood, and reduced libido can appear alongside high-stress symptoms. These are not separate problems. The cortisol system and the estrogen system are connected at the hypothalamic level, and when one shifts, the other responds.
In over 25 years of clinical practice, I have seen this pattern frequently: patients experiencing both hormonal and stress symptoms simultaneously, often told these are unrelated. They are not. If your hormonal symptoms and stress symptoms have appeared together or worsened together, that pattern is consistent with how these pathways interact. Normal stress responses are not something to fear, but understanding when a chronic pattern has begun to affect hormonal function is clinically useful.
How Estrogen Affects What Cortisol Looks Like on a Blood Test
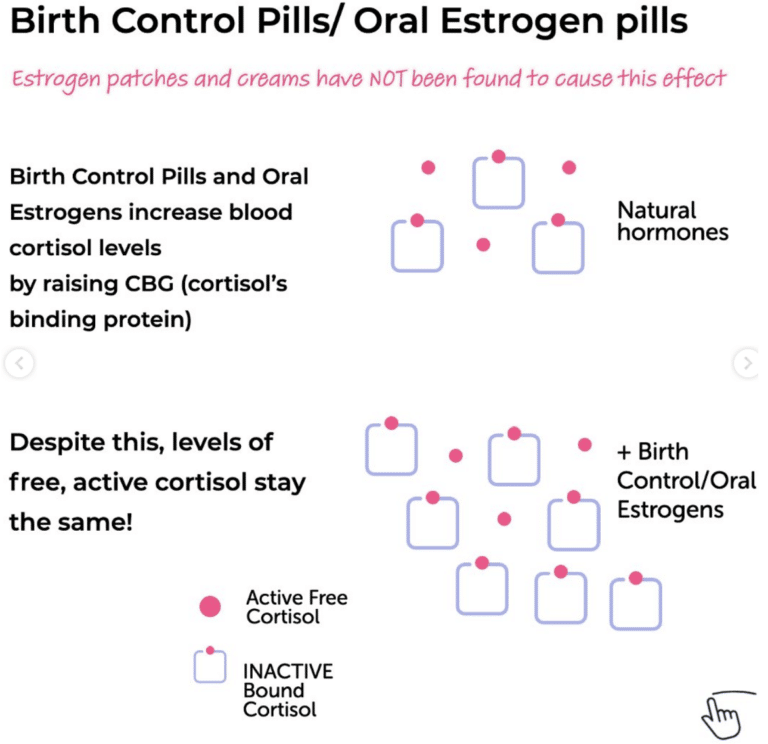
When estrogen is taken orally, as oral birth control or older forms of HRT, it passes through the liver before reaching the rest of the body. This liver first-pass metabolism stimulates production of cortisol-binding globulin (CBG), a protein that binds cortisol in the blood. One study found that oral estrogen raised total cortisol in blood by approximately 67% (1). This sounds alarming, but the critical distinction is this: total cortisol on a blood test includes both bound (inactive) cortisol and free (active) cortisol. The CBG increase raises the bound portion. Free and biologically active cortisol does not appear to be affected (3).
This means a high cortisol reading on lab work while taking oral estrogen or oral birth control may not reflect an actual increase in the cortisol that is acting on your tissues. It is a lab interpretation issue, important to understand and worth discussing with a prescriber who is aware of this pharmacokinetic effect.
| Estrogen Route | Effect on CBG / Total Cortisol | Effect on Free (Active) Cortisol |
|---|---|---|
| Oral estrogen (pills, oral HRT) | Raises CBG, so total cortisol reads ~67% higher | No significant change |
| Transdermal estradiol (patches, creams, gels) | Does not raise CBG (bypasses liver first-pass) | No significant change |
| Oral contraceptives (synthetic estrogen) | Raises CBG, similar mechanism to oral HRT | No significant change |
Cortisol and Perimenopause: Why Estrogen Volatility Changes Your Stress Response
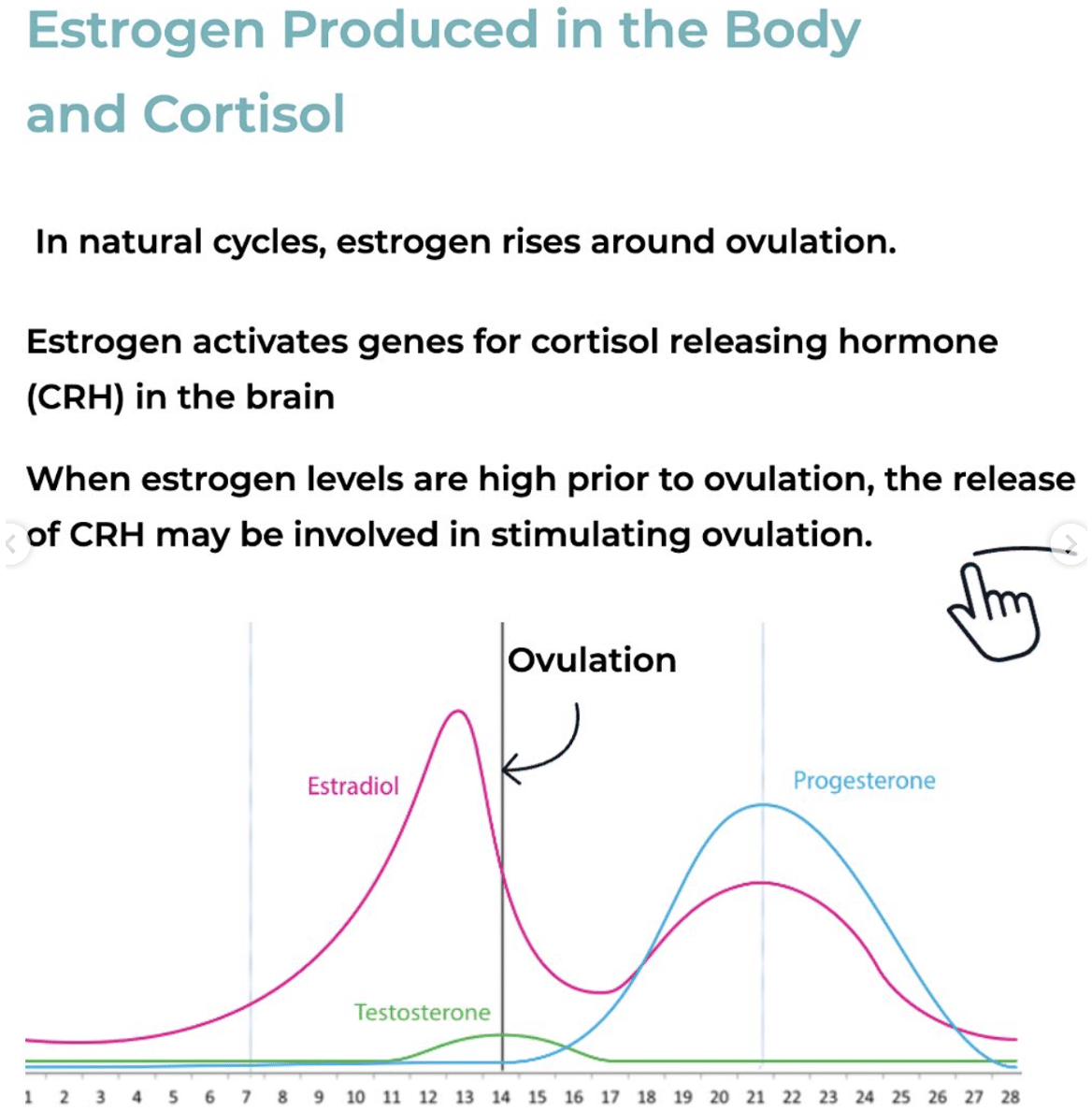
In perimenopause, estradiol does not decline steadily the way most descriptions suggest. It fluctuates sharply, with spikes that can be extremely high followed by random drops to almost zero. This volatility is the clinical reality of perimenopause, and it directly affects the cortisol system. When estradiol spikes, it activates corticotropin-releasing hormone (CRH), which stimulates cortisol release (4). This means cortisol-like symptoms such as reactivity, sleep disruption, and physical anxiety can appear in perimenopause without any corresponding increase in external stress. The trigger is internal: estrogen volatility is driving cortisol activation independently.
This produces a specific symptom pattern that many patients describe in clinical practice. It is different from ordinary stress, and it is recognizable:
- Heart racing and fearfulness that feels very physical, not like psychological worry
- Difficulty breathing, tightness, or a sensation of not getting enough air
- Feeling hot, itchy, and anxious, an internal restlessness with no clear trigger
- A “wired at night, exhausted in the morning” sleep pattern that does not respond to typical sleep strategies
- Symptom intensity that shifts day to day: some days feel manageable, others feel overwhelming
The sleep disruption in this pattern is clinically significant. Poor sleep from estrogen volatility directly impacts cortisol patterning, creating a cycle where hormonal changes worsen sleep and disrupted sleep worsens cortisol regulation. Weight changes during perimenopause can add another layer: increased adipose tissue contributes additional cortisol production from fat cells, compounding what the HPA axis is already producing.
This symptom pattern is sometimes attributed to anxiety disorder, depression, or chronic fatigue syndrome, particularly when it appears without an obvious external stressor. In clinical practice, we recognize this presentation as a pattern that frequently has a hormonal dimension. Understanding that dimension is one reason a hormonal assessment can be informative for women experiencing these symptoms during the perimenopausal transition.
If this pattern is familiar, including sleep disruption, mood variability, fatigue, or the physical anxiety described above, White Lotus Clinic’s menopause and perimenopause program offers a comprehensive hormonal assessment that includes cortisol and adrenal evaluation alongside estrogen, progesterone, and thyroid markers.
Can Estrogen Therapy Help With Cortisol Symptoms?
For many patients, estrogen therapy, particularly transdermal estradiol, addresses cortisol symptoms indirectly, through a mechanism that is more specific than “hormones help with stress.” The primary pathway is sleep. Estrogen therapy improves sleep quality. Sleep is the primary regulator of cortisol patterning and production, which is how the body resets the cortisol rhythm each day. When sleep quality improves, the cortisol pattern tends to normalize. Normalized cortisol then supports metabolic function, including reduced insulin resistance, which in turn further stabilizes cortisol regulation. This three-step cascade (estrogen → sleep → cortisol normalization → metabolic improvement) explains why estrogen therapy can help with what looks like a cortisol problem when the primary mechanism is sleep-mediated, not direct cortisol suppression.
There is also a neurotransmitter dimension. We need estrogen to make dopamine, a neurotransmitter that supports focus, motivation, and energy, and that plays a role in the sleep-wake cycle. When estrogen levels are low or volatile, dopamine production can be affected. This is why estrogen deficiency can look like mental fog, low motivation, or persistent fatigue, symptoms that overlap with depression or cognitive complaints but have a hormonal explanation. Understanding the estrogen-dopamine connection helps explain why some patients describe estrogen therapy as improving their energy and clarity, not just their hot flashes.
In clinical practice, some patients who present with fatigue, low mood, anxiety, or cognitive difficulty, and who have received diagnoses of depression, anxiety disorder, or chronic fatigue syndrome, find that estrogen therapy meaningfully changes their experience. This does not mean estrogen is universally the answer for these symptoms. It means the hormonal dimension warrants evaluation, particularly when standard approaches have not fully addressed the pattern. For patients navigating progesterone and sleep in perimenopause, this cortisol-sleep connection is also relevant: estrogen and progesterone work together in sleep regulation, and assessing both hormones in context provides a more complete clinical picture.
Cortisol and Estrogen in PCOS: The Adrenal Connection
The cortisol-estrogen relationship in PCOS involves pathways that are distinct from the perimenopause pattern, and that are often absent from standard PCOS discussions. Not all PCOS-related cortisol elevation looks the same. Some women with PCOS have elevated DHEA-S and direct HPA axis involvement, an adrenal androgen pattern where the adrenal glands are producing excess androgens alongside elevated cortisol. Other women with PCOS have high cortisol driven primarily by insulin resistance, a metabolic pathway that also disrupts the cortisol rhythm, affects sleep quality, and can increase the risk of disordered sleep. These patterns present differently, and they warrant different evaluation approaches.
What both patterns share is that high cortisol can suppress estrogen production in PCOS and impair follicle development. The biological environment for egg maturation is affected when cortisol is consistently elevated, and follicles may not develop as readily, and estrogen production from those follicles can be reduced. For women with PCOS who are experiencing both androgen symptoms and cortisol-pattern symptoms (fatigue, sleep disruption, poor stress tolerance), this intersection between the adrenal and ovarian systems is clinically significant. A comprehensive assessment that evaluates adrenal hormones alongside metabolic and reproductive markers can help clarify which factors are driving the pattern. For more on the treatment dimension of this, see our page on treating adrenal androgen excess in PCOS.
If you have PCOS and are experiencing fatigue, mood instability, or cortisol-pattern symptoms alongside androgen symptoms, a clinical assessment that includes adrenal hormone evaluation may help clarify your individual pattern. White Lotus Clinic’s PCOS program includes comprehensive hormonal and metabolic assessment.
Frequently Asked Questions: Cortisol and Estrogen
Does estrogen increase cortisol?
Oral estrogen raises cortisol-binding globulin (CBG) through liver first-pass metabolism, which causes total cortisol on blood tests to read approximately 67% higher (1). However, free and biologically active cortisol is not affected. This means a high cortisol reading while taking oral estrogen or oral birth control may reflect a binding protein change rather than an actual increase in active cortisol. Transdermal estradiol (patches, creams, gels) does not have this effect because it bypasses the liver (3).
Does estrogen lower cortisol?
Estrogen does not suppress cortisol directly. For many patients, estrogen therapy, particularly transdermal estradiol, supports cortisol regulation indirectly by improving sleep. Sleep is the primary regulator of cortisol patterning and production. When sleep quality improves, the cortisol rhythm tends to normalize, which supports metabolic function and can reduce insulin resistance. The mechanism is sleep-mediated, not direct cortisol suppression.
How does stress affect estrogen levels?
Chronic stress sustains elevated cortisol, which signals the hypothalamus to reduce release of GnRH (gonadotropin-releasing hormone). Reduced GnRH leads to lower LH and FSH, which reduces ovarian estrogen production. This is the body’s way of deprioritizing reproduction during sustained physiological threat. The result can include irregular cycles and low-estrogen symptoms alongside stress symptoms (2).
Does birth control affect cortisol levels?
Oral contraceptives containing synthetic estrogens raise cortisol-binding globulin (CBG) through the same liver first-pass mechanism as oral HRT. Total cortisol on blood tests reads higher, while free and active cortisol remains unaffected. If you receive a high cortisol result while on oral birth control, this binding protein effect is worth discussing with your prescriber.
Why is my cortisol high during perimenopause?
In perimenopause, estradiol does not decline steadily: it fluctuates sharply, with spikes that can be extremely high followed by drops to near zero. These estradiol spikes activate corticotropin-releasing hormone (CRH), which directly stimulates cortisol release independently of external stress (4). Weight changes during perimenopause can contribute further, as increased adipose tissue produces additional cortisol. This explains why cortisol symptoms can appear or worsen in perimenopause without a corresponding increase in life stress.
Can cortisol affect estrogen production in PCOS?
Yes. High cortisol can suppress estrogen production in PCOS and impair follicle development. The biological environment for egg maturation is affected when cortisol is consistently elevated. Two distinct patterns can contribute: adrenal PCOS involves elevated DHEA-S and HPA axis activation, while insulin-resistance-driven PCOS produces high cortisol through a metabolic pathway. Both can reduce estrogen production, but through different mechanisms that warrant different assessment.
Can HRT lower cortisol?
For many patients, transdermal estradiol supports cortisol regulation, primarily through sleep. Estrogen therapy improves sleep quality. Sleep regulates cortisol patterning and production. When sleep improves, the cortisol rhythm tends to normalize, supporting metabolic function and reducing insulin resistance. Estrogen is also necessary for dopamine synthesis, which affects focus, energy, and the sleep-wake cycle. The route of administration matters: transdermal estradiol does not raise CBG the way oral estrogen does, so it avoids the lab interpretation issue described above.
Should I be worried about high cortisol?
Normal stress responses are not something to fear. Cortisol is essential for energy, immune function, and recovery, and a healthy cortisol curve has natural variation throughout the day. The clinical question is whether a chronic pattern is affecting hormonal function, sleep, or metabolic health. If you are experiencing persistent symptoms alongside hormonal changes, a clinical evaluation can help clarify whether cortisol is a contributing factor in your individual situation.
How do I know if my cortisol symptoms are hormonal?
Patterns that suggest a hormonal connection include: symptoms that track with your menstrual cycle or worsen during a hormonal transition such as perimenopause; anxiety that feels distinctly physical (racing heart, difficulty breathing, heat, restlessness) rather than psychological worry; and a specific sleep pattern of feeling wired at night and exhausted in the morning. A hormonal assessment that includes cortisol, estrogen, and adrenal markers can help identify whether this pattern has a hormonal component.
What does a first consultation involve?
A first visit includes a detailed review of your symptom history, hormonal history, and relevant lifestyle factors. Based on this conversation, lab testing is typically recommended, including cortisol, estrogen, adrenal hormones, and related markers relevant to your specific pattern. A follow-up appointment reviews the findings and discusses options in the context of your individual clinical picture. A first consultation is an opportunity to understand the pattern, not a commitment to treatment.
Does cortisol affect thyroid hormones?
Yes, cortisol and thyroid hormones interact in several clinically relevant ways. Chronic cortisol elevation can affect thyroid hormone conversion and signaling, which is one reason a comprehensive assessment includes both systems. For the full explanation, see how cortisol affects thyroid hormones.
Is this the same as "adrenal fatigue"?
“Adrenal fatigue” is a widely used but contested term without a formal clinical definition. The symptoms people describe under that label, including persistent fatigue, poor stress tolerance, sleep disruption, and mood instability, are real and assessable. At White Lotus Clinic, we evaluate actual HPA axis function and adrenal hormone levels rather than relying on the adrenal fatigue framework. The goal is to identify measurable patterns, including cortisol rhythm, DHEA-S, and related markers, that can inform clinical decisions about your specific situation.
What to Expect at a First Visit
1
Initial consultation:
A detailed conversation about your symptom history, hormonal history, medical history, and the specific patterns you are experiencing, including timing, severity, and what you have already tried.
1
Lab panel recommendation:
Based on your clinical picture, relevant lab testing is recommended, typically including cortisol, estrogen, adrenal hormones (including DHEA-S), and related metabolic markers. The panel is tailored to your individual pattern, not a standard template.
1
Follow-up review:
Once lab results are available, a follow-up appointment reviews the findings and discusses clinical options in the context of your specific situation and goals.
A first consultation is an opportunity to understand your hormonal pattern, not a commitment to ongoing treatment.
Consultation fees and online booking are available on our booking page.
Meet our medical director, Dr Fiona Mcculloch, ND
Dr. Fiona McCulloch is a naturopathic doctor and the founder of White Lotus Clinic in Toronto. Her clinical practice focuses on hormonal health, including PCOS, perimenopause, menopause, and the complex interactions between the adrenal, thyroid, and reproductive hormone systems. She has been assessing and managing cortisol-estrogen interactions in clinical practice for over 25 years.
- ND: Naturopathic Doctor (Canadian College of Naturopathic Medicine)
- FABNE: Board Certified, Functional and Naturopathic Endocrinology
- Author, 8 Steps to Reverse Your PCOS
- Peer reviewer, 2023 International Evidence-Based PCOS Guidelines

Related Pages
- How cortisol affects thyroid hormones, the other side of the cortisol-hormone relationship
- DHEA-S and adrenal androgens in PCOS, covering the adrenal androgen dimension of PCOS
- Treating adrenal androgen excess in PCOS, with clinical approaches for the adrenal PCOS pattern
- Hot flashes and perimenopause treatment, covering estrogen volatility and vasomotor symptoms
- Progesterone and sleep in perimenopause, the other hormone involved in sleep regulation
- Hormone health at White Lotus Clinic, our comprehensive approach to hormonal assessment
Instagram Related Posts
View Dr Fiona’s full instagram post here!
Is a Consultation Right for You?
If the patterns described on this page, including perimenopause-related cortisol symptoms, PCOS adrenal involvement, confusion about what your cortisol lab results mean, or physical anxiety that has been attributed to stress, sound familiar, a first visit is an opportunity to review your hormonal picture with a practitioner who assesses these systems together. If you are navigating both perimenopause and PCOS, a comprehensive assessment can help clarify which factors are most relevant for your situation. A consultation is not a commitment to treatment.
References
- Wiegratz I, et al. Effect of four oral contraceptives on thyroid hormones, adrenal and blood pressure parameters. Contraception. 2003;67(5):361-366. PMID: 18045137.
- Rivier C, Rivest S. Effect of stress on the activity of the hypothalamic-pituitary-gonadal axis: peripheral and central mechanisms. Biol Reprod. 1991;45(4):523-532. PMID: 8408641.
- Bulun SE, et al. Regulation of aromatase expression in estrogen-responsive breast and uterine disease. J Clin Endocrinol Metab. 2005;90(8):4589-4594. PMID: 17492949.
- Viau V, Meaney MJ. The inhibitory effect of testosterone on hypothalamic-pituitary-adrenal responses to stress is mediated by the medial preoptic area. J Neurosci. 1996;16(5):1866-1876. PMID: 19322116.
- Chrousos GP, Gold PW. The concepts of stress and stress system disorders. Overview of physical and behavioral homeostasis. JAMA. 1992;267(9):1244-1252. PMID: 327982.
- Hale GE, et al. Endocrine features of menstrual cycles in middle and late reproductive age and the menopausal transition classified according to the Staging of Reproductive Aging Workshop (STRAW) staging system. J Clin Endocrinol Metab. 2007;92(8):3060-3067.
- Pasquali R, et al. The hypothalamic-pituitary-adrenal axis activity in obesity and the metabolic syndrome. Ann N Y Acad Sci. 2006;1083:111-128.
- Escobar-Morreale HF, et al. The molecular-genetic basis of functional hyperandrogenism and the polycystic ovary syndrome. Endocr Rev. 2005;26(2):251-282.