PCOS , Gut Peptides, Hunger and Fullness
PCOS is linked to many different hormonal imbalances. These include androgen (testosterone) excess and hormone disruptions that stem from the highest hormone-directing levels in the brain: the hypothalamus and the pituitary gland. All of these hormonal problems are made worse by insulin resistance. This is a common phenomenon in PCOS where increased insulin levels promote inflammation, and overstorage of fatty tissue around our internal organs instead of underneath the skin and its ultimately caused by androgen excess.
Patients with PCOS often report that they need to regulate their food intake or exercise to maintain good metabolic health – a common phenomenon of insulin resistance. To make this even tougher, many PCOS patients often report difficulty in detecting whether they’re full or hungry.
PCOS has also been linked to eating disorders like bulimia and binge eating disorder. We know that insulin resistance causes this problem to a degree, but is there more to learn?
Absolutely! In addition to androgens and insulin which are major problems in PCOS- there are changes in specialized hormones released by our gut. These are known as gut “neuropeptides”, and these create PCOS-related appetite imbalances which are especially problematic for those with significant insulin resistance.
These gut neuropeptides include ghrelin, cholecystokinin (CCK), GLP, Pancreatic polypeptide and peptide YY. All are crucial in regulating food intake, feelings of satiety (fullness) and hunger(1).
So how does this relate to PCOS? Prenatal exposure to androgen excess or endocrine disruption, which are likely the origin of PCOS can alter these gut neuropeptides profoundly later in life(2).
What Are Gut Peptides?

Many don’t think of the GI tract as an important endocrine organ, but it truly is.
Gut cells release a variety of orexigenic (appetite stimulating) and anorexigenic (appetite reducing) hormones. These manage our digestion, food intake, and appetite, and also regulate metabolism. Specific types of enteroendocrine cells (hormone-releasing cells) release gut peptides as a reaction to food intake.
In addition to these gut-based secretions, common brain neurotransmitters such as dopamine, 5-HT, and Norepinephrine also tweak our sensations of hunger and satiety. And all these signals act in concert together across the brain!
Ghrelin: The Hunger Hormone
The stomach contains cells that secrete ghrelin. This is a peptide, and the only appetite-stimulating hormone that circulates in the blood. Ghrelin spikes before meal times and travels to the brain. It crosses the blood-brain-barrier and makes us feel hunger sensations. This is why it’s often called the “hunger hormone”
Once it arrives, ghrelin acts on the hypothalamus to boost appetite!
Ghrelin level changes are linked to elevated androgen levels in PCOS. In a healthy metabolism, ghrelin should reduce with food intake or a glucose load, suppressing appetite. For those with this condition, this is blunted—possibly causing an inability to tell when one is “full”. (2)
CCK, GLP: Fullness Signals
The small intestine contains specialized cells that produce cholecystekinin: an fullness-signalling peptide. CCK was the first appetite-related gut hormone discovered, and is also found in the brain.
Those with PCOS have lower meal-stimulated CCK responses. This again cases a lack of ability to feel “fullness”, unlike how those with normal levels may feel after eating a meal. Although this was more marked in those who had higher body mass indices, another trial found that this also applied to lean patients with PCOS. Furthermore, the lean patients’ appetite and fullness signals actually related more to their increased testosterone levels. (3)
L cells in the small and large intestines and produce glucagon-like peptide (GLP)-1/2. This is a strong appetite regulator, and also governs feelings of fullness. GLP-1 secretion is known to be impaired in insulin resistance, so this will also apply to insulin-resistant PCOS.
Basically, signals for fullness are less powerful in PCOS.
Brain-Gut Communicators : The NPY Peptides
The members of the NPY peptide family include Neuropeptide Y (NPY), Protein YY (PYY) and pancreatic polypeptide (PP). These are found at various levels of the gut–brain axis.
These peptides play important roles in gut-brain communication. They regulate food intake, energy homeostasis, and even anxiety, mood, and stress resilience.
After a meal, and PP and PYY are released from intestinal L cells in proportion to amount of energy consumed. They slow food’s passage through the gut, inhibiting further intake, and creating a sense of “fullness”. It’s thought that PP is also able to signal fullness to the hypothalamus brain region, and may even cross the blood-brain barrier. (4)
Whereas PP and PYY are primarily found in the gut, NPY is the most abundant neuropeptide in the brain. It’s found in areas ranging from the brainstem to the hypothalamus, and the cerebral cortex (white matter). NPY also seems to play a role in pain, mood, and stress coping.
Insulin resistance causes irregular secretion of these gut neuropeptides, creating difficulty in sensing the difference between fullness and hunger. (1)
Satiety : A Critical Factor to Thriving with PCOS
So, now that we’ve gone through all of the science behind WHY patients with PCOS have troubles with dysregulated gut peptides – let’s talk about why that is a problem? Of course we know that it can cause us to eat more than we need, increasing insulin resistance. That part is obvious – but what else happens when you never feel “full”?
What is Satiety and Why Does It Matter?
<p>Satiety is that feeling of satisfaction after you eat – where your mind no longer ruminates on getting more food. We all know that when hunger is present, it’s very difficult to focus on anything else. Of course we eat for enjoyment, but a fairly large part of why we eat is to remove that annoying feeling of hunger that takes up so much space in our thoughts. Satiety allows us to “move on” and not feel the sense of hunger overpowering our thoughts. To say the least, the lack of satiety is unsettling, stressful, and takes up a large portion of our brainpower.</p>
Diets and Food Restriction Only Make PCOS Worse
We know that many patients with PCOS spend years in various states of dietary restriction – often depriving themselves of food in an attempt to lose weight. Many end up bingeing after periods of restriction – leading to blood sugar dysregulation, depression and anxiety around “failures” at controlling food intake. This is SO harmful!
When these gut peptides are already dysregulated by androgen excess and insulin resistance in PCOS and a sense of satiety isn’t possible with a regular meal- it promotes a huge array of problems. It’s no wonder – imbalances of these neuropeptides are associated directly with stress, anxiety, depression, and other mood-related disorders. These neuropeptides can have direct effects on mood, acting like neurotransmitters, as well as indirect ones – affecting the microbiome in ways that change the microbiome – gut- brain axis.
Improving Gut Peptide Balance in PCOS
What we eat has a huge impact on gut-brain balance. Eating foods that are good for our microbiome help these beneficial bacteria secrete short-chain fatty acids that can positively influence the behaviour of the specialized hormone-secreting cells in our gut. In addition to this, the balance and composition of nutrients in what we eat can affect neuropeptides and our sense of satiety very much.
Satiety: How to Promote a Sense of Fullness and Satisfaction in PCOS
Healthy, balanced and sustainable nutrition
Minimize sugar, as well as refined, low-quality foods. Include a variety of vegetables and fibres—these will feed the microbiome and help shift it to a metabolically beneficial type. Meals should be composed of protein, healthy fat, non-starchy vegetables and a source of low glycemic index, or high resistant starch carbohydrates. Resistant starch such as green banana flour, potato starch, cooked and cooled rice and fibre from legumes are excellent fuel for our microbiome.
Improve your natural satiety signals
Improve your natural satiety signals as much as possible with meal structures that promote feelings of fullness. The best components of a meal to support satiety include protein, healthy fat, and fibre. Always combine carbohydrates with these macronutrients to reduce blood sugar rollercoaster effects.
Avoid restrictive “diets”
Avoid restrictive “diets” and meal plans. These often trigger stress, feelings of unrest, and ultimately bingeing. In androgen excess, our gut neuropeptides are already in a state of dysregulation and these types of diets can only make matters worse particularly over the long haul. Avoid looking at foods as “bad” and “good” but rather focus on how to create a sense of satiety with whole, healthful foods.
Good digestive hygiene
Good digestive hygiene is important for proper digestion. Don’t eat while engaging in other activities such as work, or while distracted. Chew properly and don’t eat too quickly—take time to enjoy your meal. These improve satiety signals and allow for maximum feelings of fullness.
Glucomannan fibre
From Konjac root promotes satiety. Konjac “rice” “noodles” and fibre supplements are a healthy and safe way to increase sensations of fullness. Glucomannan is also an excellent fibre that supports the microbiome. Other soluble fibres can also provide similar benefits. A high fibre diet is not only satisfying but promotes optimal gut neuropeptide secretion.
Probiotics
A variety of probiotic strains have been shown to impact the metabolism positively. In contrast, lower bacterial diversity and a higher ratio of the firmicutes division negatively affects metabolic health. Always consult with your healthcare practitioner, as different strains have many different effects. In fact, some may actually have pro-inflammatory effects. A few strains found to be specifically supportive to metabolic health include Lactobacillus plantarum, Lactobacillus rhamnosus, Lactobacillus paracasei, Bifidobacterium M13-4, Bifidobacterium animalis subsp. Lactis, Saccharomyces boullardi, and akkermansia muciniphilia, Bacillus indicus, Bacillus subtilis, Bacillus coagnulins and others.
Address Stress and Work-Life Balance
Stress has repeatedly been found to dysregulate our satiety signals. In situations of anxiety, or chronic stress people feel full less easily and will spontaneously eat more. It’s been found that satiety is decreased while consuming food while working on a computer – a situation that is common during the pandemic. To work on stress practice mindfulness, yoga, meditation, take a bath or find literally any stress reliving activity you enjoy. One herbal supplement that can be helpful for both stress and satiety is Relora (a combination of magnolia and phellodendron).
Get sufficient sleep
It’s been found that decreased sleep quality or quantity will decrease satiety signals and cause us to consume more food throughout the day.
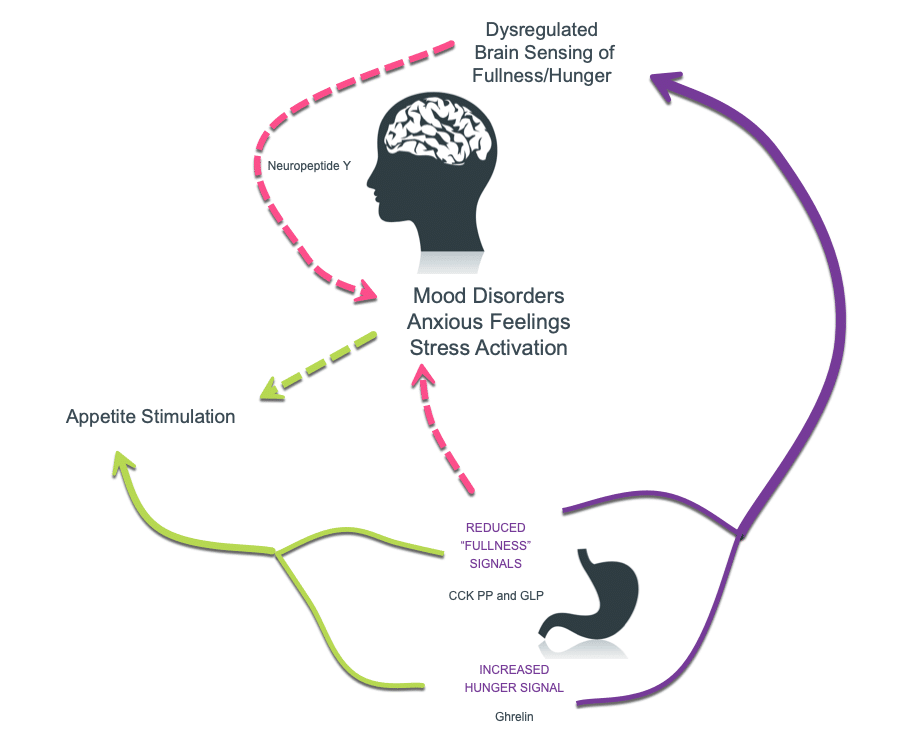
Adapted from: Ilie, I. R. (2020). Neurotransmitter, neuropeptide and gut peptide profile in PCOS-pathways contributing to the pathophysiology, food intake and psychiatric manifestations of PCOS. In Advances in Clinical Chemistry (Vol. 96, pp. 85–135). Academic Press Inc.
References
- Ilie, I. R. (2020). Neurotransmitter, neuropeptide and gut peptide profile in PCOS-pathways contributing to the pathophysiology, food intake and psychiatric manifestations of PCOS. In Advances in Clinical Chemistry (Vol. 96, pp. 85–135). Academic Press Inc.
- Abbott, D. H. (2017). Neuronal androgen receptor: Molecular gateway to polycystic ovary syndrome? Proceedings of the National Academy of Sciences of the United States of America, 114(16), 4045–4047.
- 3, 4. Barber, T. M., Casanueva, F. F., Karpe, F., Lage, M., Franks, S., McCarthy, M. I., & Wass, J. A. H. (2008). Ghrelin levels are suppressed and show a blunted response to oral glucose in women with polycystic ovary syndrome. European Journal of Endocrinology, 158(4), 511–516.
- Holzer, P., Reichmann, F., & Farzi, A. (2012, December). Neuropeptide Y, peptide YY and pancreatic polypeptide in the gut-brain axis. Neuropeptides. Neuropeptides.
- Liu, R., Zhang, C., Shi, Y., Zhang, F., Li, L., Wang, X., … Ding, X. (2017). Dysbiosis of gut microbiota associated with clinical parameters in polycystic ovary syndrome. Frontiers in Microbiology, 8(FEB).